Recently, the "Solid-State NMR and Heterogeneous Catalysis" team from the Innovation Academy for Precision Measurement Science and Technology (APM) was invited by Accounts of Chemical Research, an authorative review journal of the American Chemical Society, to write a topical review paper titled "Intermolecular interactions in zeolite catalysis: insights from solid-state NMR spectroscopy." This journal is known for publishing systematic research findings by authors. The article comprehensively summarizes the significant research progress made by the team in recent years in developing and applying solid-state nuclear magnetic resonance (NMR) techniques to reveal intermolecular interactions in zeolite catalysis.
Zeolites are the most widely used catalytic materials in petrochemical production. The confinement effect within their nanopores significantly amplifies the weak intermolecular interactions between host-guest, guest-guest, and environmental effects (such as water), thereby profoundly influencing the reaction pathways, selectivity, and catalyst lifespan. However, these interactions are difficult to capture and quantify experimentally, and their specific mechanisms and regulatory patterns have always been major challenges in this field. In recent years, the research team has made a series of important breakthroughs in the field of high-resolution, high-sensitivity solid-state NMR methodology and successfully applied them to the study of zeolite catalytic systems.
In terms of studying host-guest interactions within zeolite pores, the team established double-resonance solid-state NMR methods such as ¹³C-{²⁷Al} 、 ¹H-{⁹⁵Mo}, enabling precise detection of the spatial proximity between organic molecules and zeolite active sites for the first time. Using this technology, researchers characterized the microscopic structure of "supramolecular active centers" formed by Brønsted acid sites and hydrocarbon pool species in zeolites and directly identified the key molybdenum active species and their evolutionary pathways in the methane dehydroaromatization reaction.
To address the complex guest-guest interactions in reaction networks, the team developed a new solid-state NMR method based on two-dimensional ¹³C-¹³C correlation spectroscopy, successfully elucidating the non-covalent interactions between cyclic carbocation intermediates and co-adsorbed molecules in the methanol-to-olefins reaction, including cation-dipole and cation-π,and other forms. This discovery clarified the key mechanisms of weak interactions in regulating product selectivity and molecular sieve carbon deposition deactivation.
Furthermore, by combining solid-state NMR with magnetic resonance imaging techniques, the team revealed the regulatory effects of water solvents in the reaction environment. The study found that the "micro-hydrophobic effect" induced by water molecules significantly promotes the binding of benzene to surface methoxy species and clarified the differences in hydrophilicity/hydrophobicity between confined carbocations and aromatic species, providing new insights into the regulatory mechanisms of reaction activity and product selectivity.
Professor WANG Chao is the first author of the article, and Professor XU Jun is the corresponding author.
This series of research work was supported by the National Natural Science Foundation of China, the Chinese Academy of Sciences, and Department of Science and Technology of Hubei Province.
Link to the article: https://pubs.acs.org/doi/10.1021/acs.accounts.5c00652
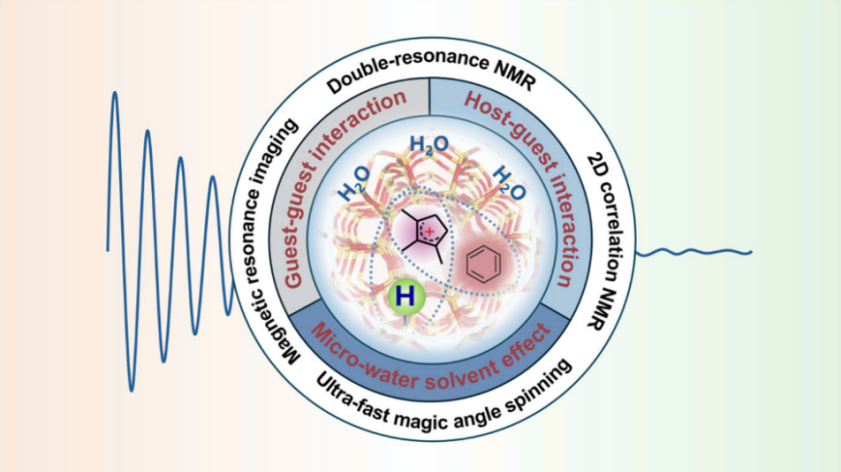
Solid-State NMR Studies of Intermolecular Interactions in Zeolite Catalysis
