Recently, the Solid-State NMR and Heterogeneous Catalysis Team at the Innovation Academy for Precision Measurement Science and Technology (APM) has made significant progress in the research on intermediates of catalytic reactions in zeolite molecular sieves. The research team has revealed for the first time that trace molecular oxygen can significantly alter the reaction pathway of carbocations, which are key reaction intermediates in the confined environment of zeolite pores, thereby regulating the selectivity of important catalytic reactions such as methanol-to-hydrocarbon conversion and the catalyst lifespan. The relevant research results have been published in the Journal of the American Chemical Society.
Carbocations are key reaction intermediates in the hydrocarbon conversion catalyzed by zeolites. Their high reactivity and transient characteristics make the direct characterization and understanding of their reaction behavior highly challenging. During the zeolite catalytic process, trace oxygen is often present in the feedstock or during the catalyst regeneration process, but the mechanism of its action on catalytic reactions has remained unclear.
The research team combined multiple technical approaches, including in-situ solid-state NMR spectroscopy, in-situ UV-Vis spectroscopy, and online mass spectrometry, to systematically investigate the reaction behavior of carbocation intermediates on ZSM-5 zeolites in an oxygen-containing atmosphere (Figure 1a, b). The study found that even trace oxygen at the ppm level can significantly promote the conversion of cyclopentenyl carbocations. Oxygen can cause cyclopentenyl carbocations to undergo hydroxylation, which is then converted into cyclopentenone species, and further undergo aromatization reactions to produce aromatic hydrocarbons (Figure 1c). Through the isotope-labeling experiment using ¹⁷O₂, researchers directly confirmed via ¹⁷O NMR that oxygen atoms from molecular oxygen had entered the carbonyl group of cyclopentenone, providing direct evidence for the oxidation mechanism. This study has revealed the dual roles of oxygen in molecular - level catalytic reactions: on one hand, it promotes the formation of aromatic hydrocarbon products by oxidizing carbocation intermediates, altering the selectivity distribution of olefins/aromatic hydrocarbons; on the other hand, the oxidation reactions also accelerate the generation of carbon-deposit precursors such as polycyclic aromatic hydrocarbons, thus speeding up the deactivation of the catalyst. This oxidation mechanism is equally applicable to reactions such as ethylene conversion, indicating that oxygen has a universal regulatory effect on the reactivity of carbocations.
This study provides brand-new molecular-level insights into the regulatory mechanism of trace oxidants on reaction pathways in practical catalytic systems, and offers new strategies for optimizing the performance of catalysts (such as product selectivity and stability).
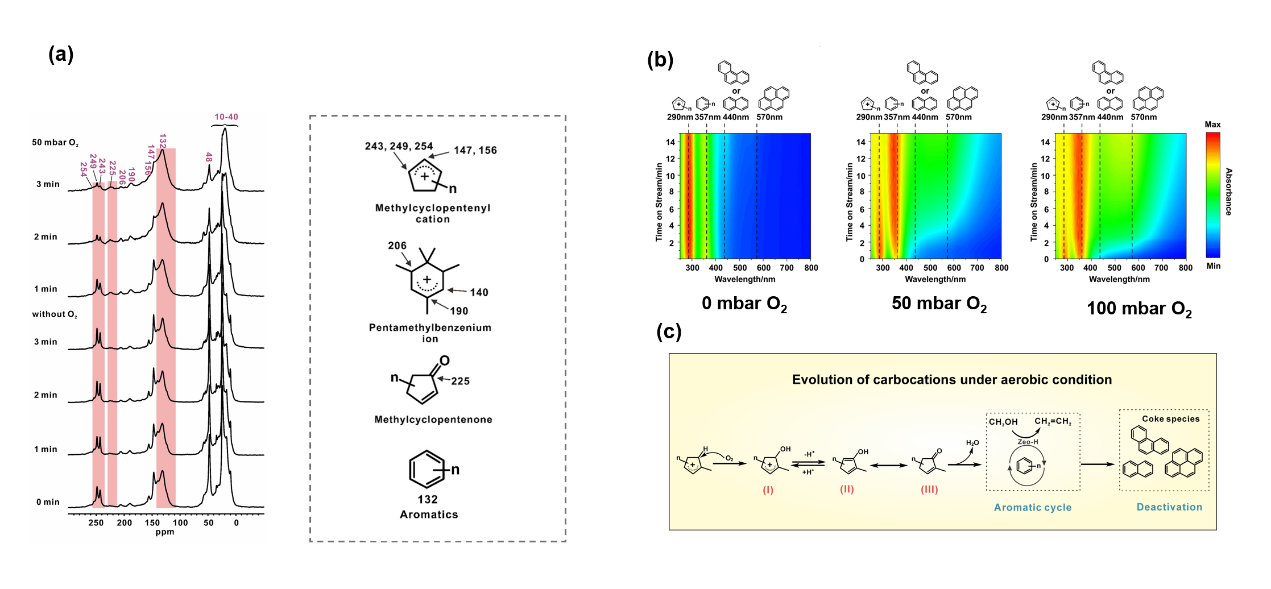
(a) In-situ ¹³C MAS NMR spectra of the evolution of carbenium ions on molecular sieves under oxygen-containing/oxygen-free atmospheres; (b) In-situ UV-Vis spectra of the evolution of carbenium ions under different oxygen partial pressure conditions; (c) The reaction mechanism of molecular oxygen oxidizing carbenium ions
The relevant research was published in the Journal of the American Chemical Society under the title "O2-Mediated Reactivity of Confined Carbenium Ions in Zeolites". CAI Wenjin, a doctoral candidate from APM, is the first author of the paper, while Professor WANG Chao and Professor XU Jun from the same academy are the corresponding authors of the article.
This research work has received support from the National Key Research and Development Program, the National Natural Science Foundation of China, and the Chinese Academy of Sciences.
Link to the article: https://pubs.acs.org/doi/10.1021/jacs.5c20563
