Recently, the research team from the Innovation Academy for Precision Measurement Science and Technology (APM) has made new progress in the research on nuclear magnetic resonance (NMR) spectroscopy strategies of "separating spectral peaks without separating samples". A phenylalanine-lysine oligopeptide (FK)₄ has been developed, which can self-assemble to form a nuclear magnetic resonance (NMR) medium with bifunctional properties. This medium enables the simultaneous measurement of enantiomer discrimination and relative configuration analysis within the same system, providing a new approach for the stereochemical structure analysis of organic compounds. The relevant research results have been published in the Journal of the American Chemical Society.
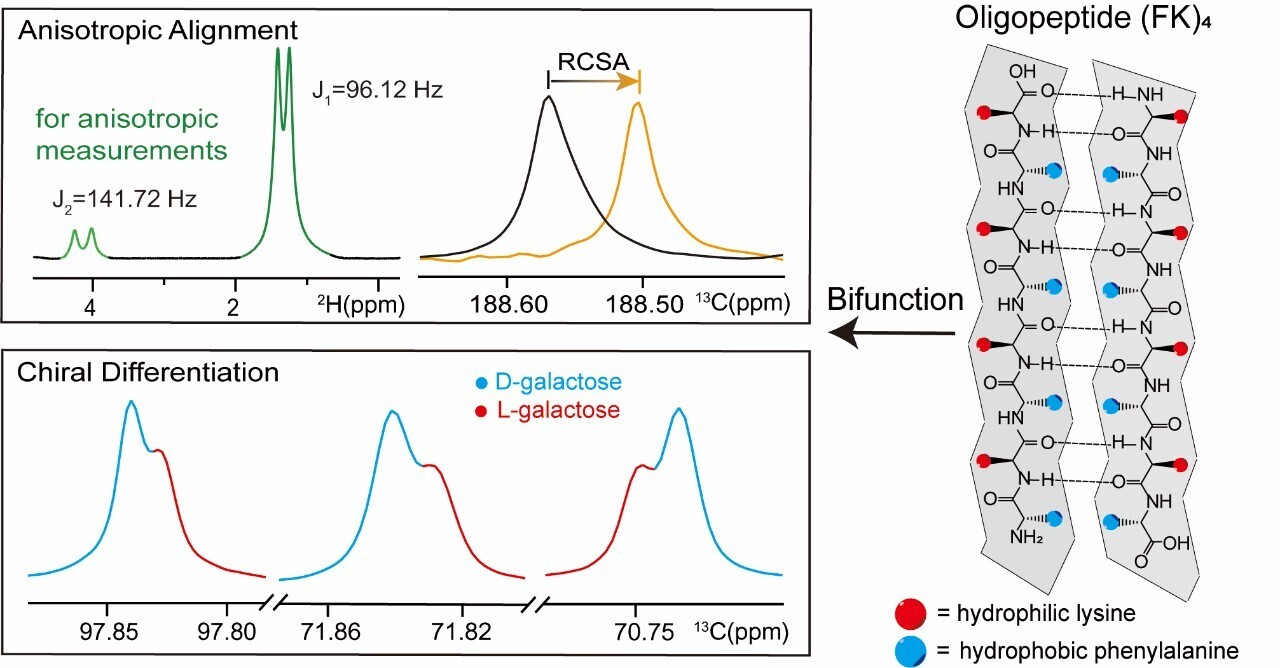
Figure 1. Characterization of the bifunctional properties of (FK)₄. Image source: J. Am. Chem. Soc.
In the process of stereochemical structure analysis of organic molecules, chiral analysis and anisotropy measurement are usually carried out step-by-step relying on different experimental systems, resulting in a cumbersome operational procedure. In response to this long - standing technical bottleneck, the research team started from the (FK)₄ self-assembly system and developed a novel alignment medium (FK)₄. (FK)₄ can self-assemble in solution to form a lyotropic liquid-crystal phase with a β-sheet nanofiber structure. On one hand, it provides a weak-orientation environment for small molecules, which is used to measure residual chemical shift anisotropy (RCSA) for the analysis of relative configuration. On the other hand, the differential interactions between its chiral polypeptide backbone and enantiomers can induce the splitting of enantiomer NMR signals, enabling high-resolution chiral discrimination. Thus, the dual-function integration of relative configuration analysis and enantiomer recognition has been simultaneously achieved within the same system. The experiments indicate that in proton-decoupled ¹³C NMR spectra, amino acids, carbohydrates, and chiral drug molecules all exhibit clear enantiomer signal splitting. The integration results show a good linear relationship with the HPLC reference data, verifying their quantitative reliability. This system is simultaneously compatible with ¹H and ¹⁹F nuclear detection. It can also self-assemble to form a liquid-crystal phase in protic organic solvents such as methanol and isopropanol. The degree of alignment can be regulated by temperature, enabling the controllable switching of anisotropy intensity. This study expands the anisotropy NMR measurement strategies, integrating structural analysis and chiral analysis to be completed under a single sample and unified experimental conditions.
On this basis, the research team established a "plug-and-play" analytical process: First, the reaction products are directly dissolved in deuterated methanol containing (FK)₄, and enantiomer signal splitting can be observed via ¹³C NMR. Subsequently, temperature regulation is carried out on the same sample to achieve reversible conversion between the anisotropic state and the isotropic state. By measuring the chemical shift difference (ΔΔRCSA) in these two states and combining it with computational chemistry fitting analysis, the relative configuration of the products can be accurately determined. This method enables the simultaneous completion of chiral recognition and relative configuration analysis in the same sample. It provides new ideas for the application of anisotropic NMR technology in stereochemical research and lays a methodological foundation for absolute configuration analysis.
The research-related findings were published in the Journal of the American Chemical Society under the title "A Bifunctional Phenylalanine-Lysine Oligopeptide Enabling Both Chiral Differentiation and Relative Configuration Analysis by Nuclear Magnetic Resonance Spectroscopy". Postdoctoral fellow PENG Yun and doctoral student ZHU Mingjun from APM are the co-first authors of the article, while researchers HE Lichun and LIU Maili are the co-corresponding authors.
This research work was supported by the B-class Pilot Project of the Chinese Academy of Sciences and the National Natural Science Foundation of China.
Link to the article: https://doi.org/10.1021/jacs.5c22161
