Recently, the Solid-State NMR and Heterogeneous Catalysis research group at the Innovation Academy for Precision Measurement Science and Technology (APM) made progress in developing solid-state NMR methods to resolve the interfacial acid site structures of catalysts. Utilizing self-developed two-dimensional (2D) spectral-editing solid-state nuclear magnetic resonance (NMR) methods, the researchers discovered interfacial pseudobridging silanol (PBS) pair active centers with Brønsted acidity in hydrothermally treated ASA catalysts. Furthermore, the study revealed the microstructure and formation mechanism of these ASA catalysts. The findings were recently published in the prestigious Journal of the American Chemical Society.
Amorphous silica-alumina (ASA) is a crucial solid acid catalyst. It is widely used in fields such as petroleum catalytic cracking and biomass conversion due to its low cost and tunable acidity. However, the exact atomic structure of its Brønsted acid sites (BAS)—whether classic bridging hydroxyls or pseudobridging silanols—has remained controversial. Conventional characterization techniques struggle to resolve these microscopic interfacial hydroxyls due to structural disorder and complexity. These methods are either insensitive to hydrogen (e.g., electron microscopy), lack spatially localized information (e.g., infrared spectroscopy), or are restricted to external surface probing (e.g., X-ray photoelectron spectroscopy). Solid-state NMR is an effective tool for probing atomic-level local structures in amorphous systems. Nevertheless, existing NMR techniques face severe challenges in characterizing interfacial hydroxyls. While Dynamic Nuclear Polarization (DNP) enhances sensitivity, signals from polarizing solvents often interfere with the direct observation of ¹H signals. Furthermore, in conventional ¹H 2D NMR experiments, the sparse signals of acidic sites suffer from severe spectral overlap with abundant non-acidic hydroxyls (Si-OH, Al-OH) and hydrogen-bonded networks. This overlap significantly limits the precise resolution of catalytic active sites in ASA catalysts.
In this work, the researchers first employed 13C MAS NMR experiments with acetone adsorption to confirm that hydrothermal post-treatment significantly increases the density of Brønsted acid sites in ASA catalysts. To investigate the structural origins of this acidity enhancement, one‑dimensional (1D) 29Si MAS NMR and 2D 27Al MQMAS experiments were subsequently conducted. They discovered that after high-temperature hydrothermal treatment, the penta-coordinated aluminum (Al(V)) on the alumina surface diminishes. Concurrently, the structure of tetra-coordinated Al(IV) species progressively evolves from Al-[O-Al]4 toward Al-[O-Si]n (n = 1–4). This structural evolution proves that high-temperature hydrothermal treatment promotes the integration of SiO2 and Al2O3 domains, generating local interfaces. By leveraging Dynamic Nuclear Polarization Surface Enhanced NMR Spectroscopy (DNP-SENS), the detection sensitivity for these interfacial species was increased by nearly two orders of magnitude. Furthermore, by applying the group's previously developed PT-J/D-HMQC method, the researchers obtained 2D 29Si-27Al heteronuclear correlation data at the ASA interface at natural isotopic abundance. This clarified that the formation of pseudobridging silanols during post-treatment is the primary driver for the increased Brønsted acidity.
To obtain a more refined structure of the interfacial acidic hydroxyls, the researchers applied their self-developed 27Al-filtered (edited) 2D high-resolution 1H-1H homonuclear correlation (f-DQ/SQ) method. This technique enabled the selective observation of interfacial hydroxyls in ASA. It effectively eliminated interference from non-acidic hydroxyls on the alumina or silica surfaces. A key spectral peak at (1.8, 3.6) ppm was assigned to pseudobridging silanol pairs situated close to aluminum. This confirms the massive formation of PBS pairs during the post-treatment process. Finally, the researchers measured the precise distances between the hydrogen and aluminum atoms within these PBS pairs. They achieved this using ²⁷Al-filtered 1H-1H f-DQ/SQ, 1H-{27Al}1 S-RESPDOR experiments, and spin dynamics simulations. By combining these measurements with Density Functional Theory (DFT) calculations, they successfully determined the optimal structure of the PBS pairs. This specific structure, located at the SiO2 and Al2O3 interface, acts as a crucial source of acidity in ASA catalysts. These findings reveal the microscopic structure, distribution, and transformation of interfacial species in amorphous silica-alumina at the atomic and molecular levels. This insight is of great significance for understanding catalytic acid properties and guiding future acidity modulation.
The research findings were published in the Journal of the American Chemical Society under the title "Interfacial Brønsted Acid Site Architectures on Amorphous Silica-Alumina Resolved by Heteronuclear-Filtered Two-Dimensional Solid-State NMR Correlation Spectroscopy". ZHENG Mingji, a doctoral student at APM, and Dr. ZENG Shuangqin from the SINOPEC Research Institute of Petroleum Processing are the co-first authors of the paper. Professor WANG Qiang, XU Jun, and DENG Feng serve as the co-corresponding authors. Professor CHRISTOPHE Copéret from ETH Zurich, Switzerland, also participated in the collaborative discussions for this work.
This research was supported by the Ministry of Science and Technology, the National Natural Science Foundation of China, the Chinese Academy of Sciences, and the Hubei Provincial Department of Science and Technology.
Link to the article: https://pubs.acs.org/doi/full/10.1021/jacs.5c20339
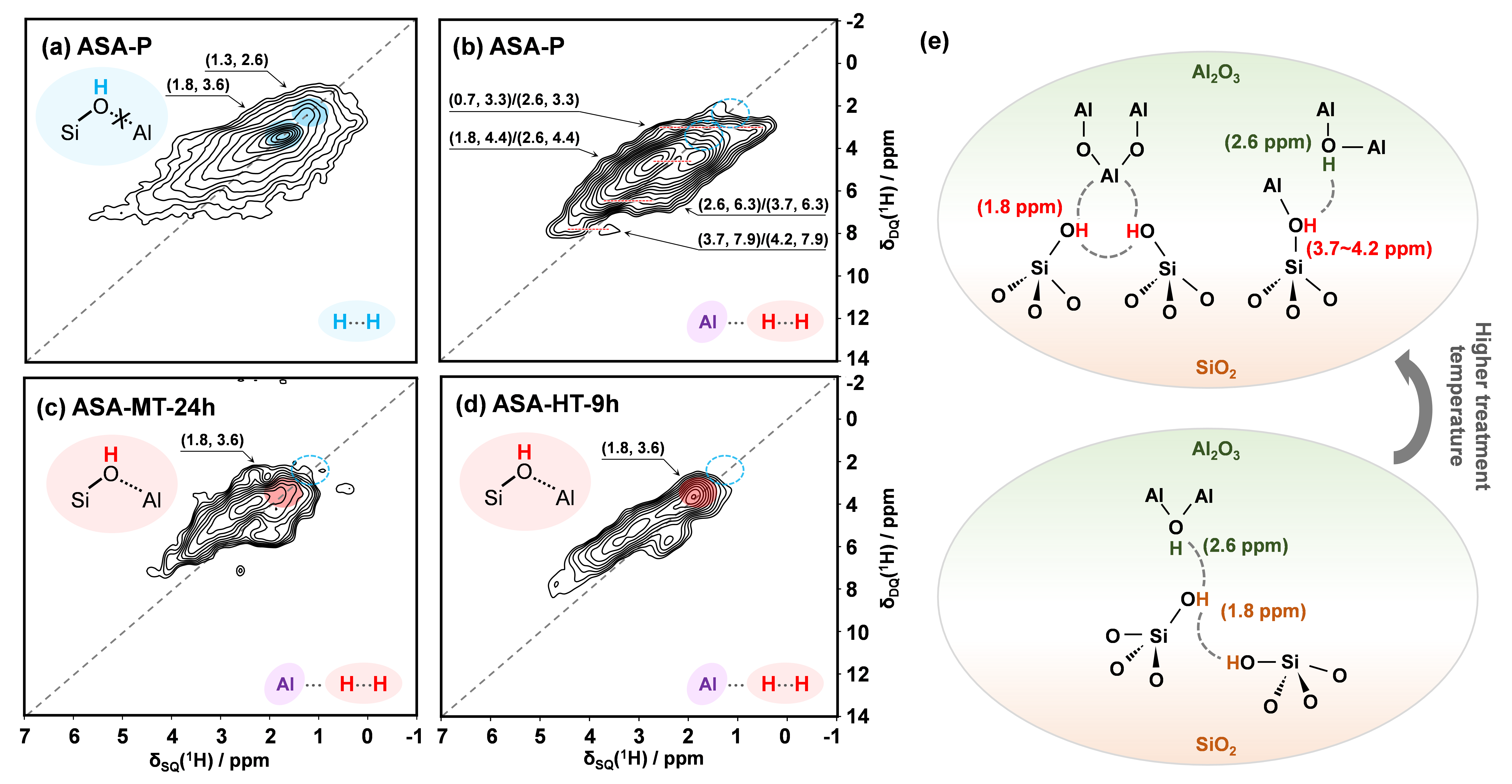
Figure 1. (a) Two-dimensional 1H-1H DQ/SQ NMR spectrum of dehydrated ASA-P; (b-d) Two-dimensional 1H-1H f-DQ/SQ NMR spectra of dehydrated ASA-P, ASA-MT-24h, and ASA-HT-9h; (e) Proposed structural model of ASA surface species
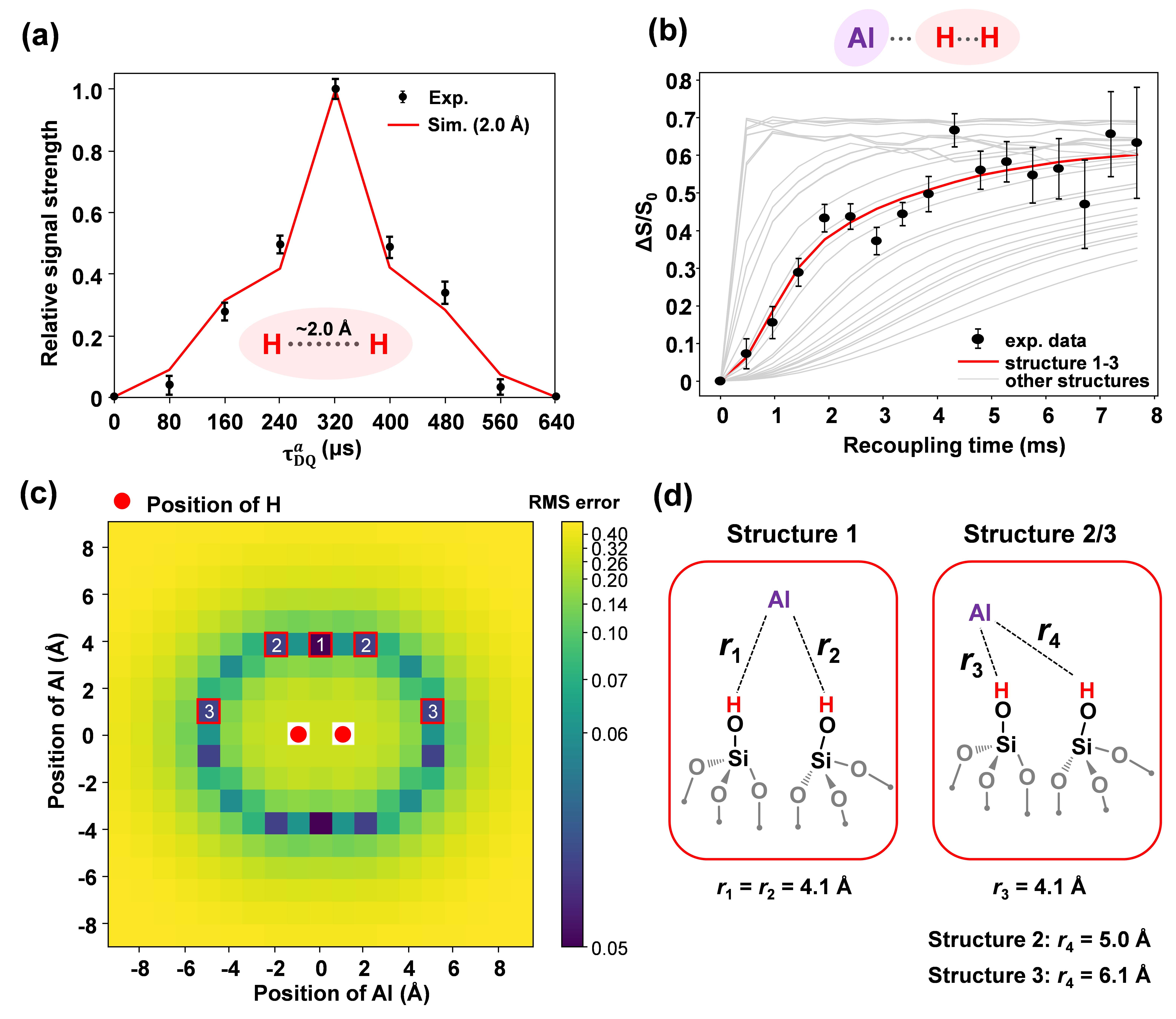
Figure 2. Measurement of the inter-nuclear distance of "pseudo-bridging hydroxyl pairs" in hydrothermal-treated ASA catalysts; (a) Constant-time evolution 1H-1H f-DQ/SQ experiments and fitting; (b) 1H-{27Al} S-RESPDOR experiments and fitting; (c) Root-mean-square error between the experimental data of 1H-{27Al} S-RESPDOR and the results of spin dynamics simulations; (d) Two most likely structural models of pseudo-bridging hydroxyl pairs proposed based on NMR/DFT results
